About RainMed
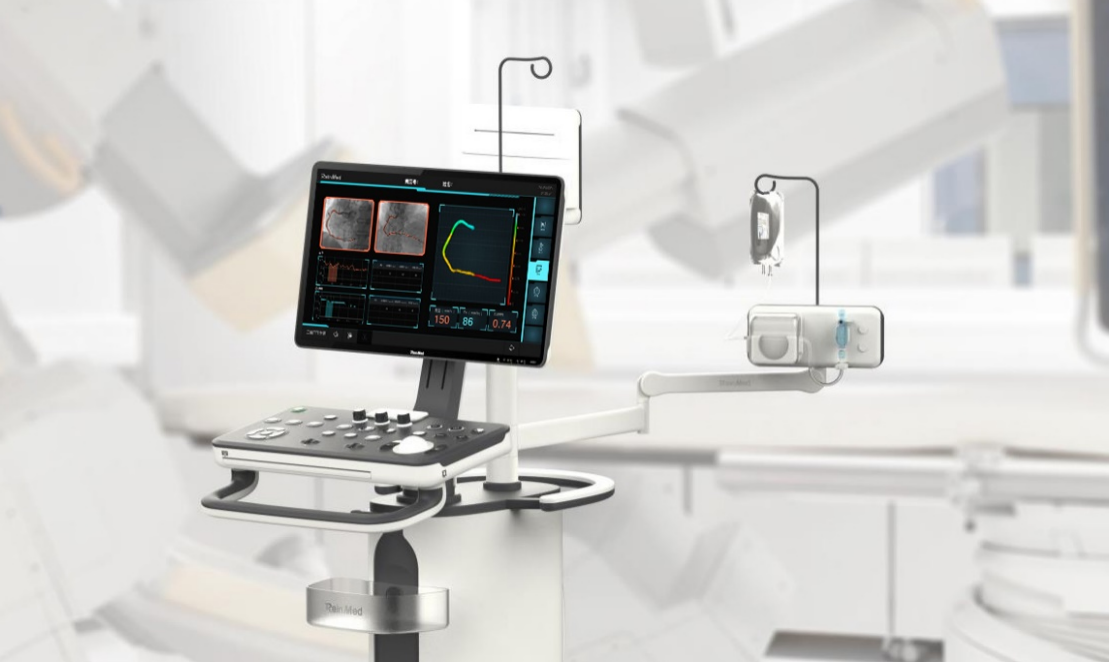
RainMed Medical Group, founded in 2014, is a national high-tech enterprise committed to developing world's leading vascular interventional surgical robot. Currently, it is focusing on the design, development and commercialization of digital functional diagnosis module caFFR system, caIMR system and FlashBot vascular interventional surgery robot. With independent innovation and great efforts, RainMed Medical has gradually built up a global R&D, production, sales and service network.
RainMed Medical has actively cooperated with top medical centers and experts at home and abroad to build a perfect patent system. A great number of research results on caFFR and caIMR have been continuously published in authoritative journals such as Cardiovascular Research and JACC: Cardiovascular Interventions. In addition, RainMed has signed strategic cooperation agreements with top domestic medical device distribution service platforms, developed a strict quality control system and a sound supply process, and successfully passed the medical device quality management system certification......
Recent News
More information
Welcome to RainMed
Caring for employees’ work and life
Provide a platform for employee growth and promotion
CONTACT US
0512-62622215
Address: Building 31, Northeast District, Nano City, No. 99 Jinji Lake Avenue, Suzhou industrial park
E-mail: market@rainmed.com

WeChat public account

Recruitment QR Code
Page Copyright© 2021- Suzhou Rainmed Medical Technology Co., Ltd. 苏公网安备32059002003601号 (苏)-非经营性-2021-0149